Hyperpolarized MRI
The emerging MRS technique of dynamic nuclear polarization (DNP, or "hyperpolarization") dramatically enhances the observed signals of 13C-labeled metabolic substrates, allowing real-time noninvasive imaging of both the injected substrate and downstream metabolic products. Within the constraints of high polarizability, low toxicity, rapid cellular uptake and metabolism, sufficient chemical shift separation between products, and a T1-limited observation window, many 13C-labeled candidates have been investigated for in vivo measurement of key metabolic pathways. A polarizer externally aligns magnetic moments of the substrate 13C nuclei prior to injecting the polarized substrate into the subject as bolus.
Recognizing the significant potential of this new technology, UT Southwestern created a Hyperpolarization Imaging Center dedicated to developing state-of-the-art imaging techniques. Funded primarily by multiple awards from the National Institute of Health (NIH), UT Southwestern purchased a clinical MRI scanner (GE 3T Discovery 750w, Grant #S10RR029119) and a clinical DNP polarizer (GE SPINlab™, Grant #S10OD018468) dedicated to this project. UTSW installed the MRI scanner and the SPINlab™ in adjacent spaces convenient for clinical research activity and patient care. The AIRC also offers a new preclinical DNP polarizer (Polarize SpinAligner, Grant #S10OD028490) and a para-H2 generator (Bruker).
Probe Development
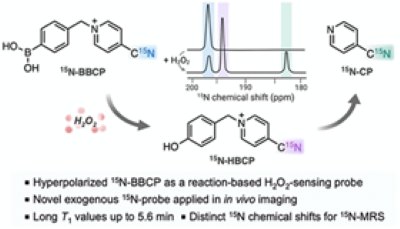
Hyperpolarized probe development at the AIRC focuses on two main areas: metabolic probes and hyperpolarized sensors. Hyperpolarized 13C-labeled metabolic substrates include [3,4-13C2, 2,3,4,6,6-2H5]glucose and [1-13C]glycerate, D-[1-13C]gluconolactone, diethyl [1,2-13C2]-2-ketoglutarate, [1-13C]-2-ketoisocaproate, and [1,3-13C2]acetoacetate to monitor glycolysis, the pentose phosphate pathway, the TCA cycle, branched chain amino acid metabolism, and beta-oxidation, respectively. AIRC teams designed ghyperpolarized MR sensors for various physiological parameters (pH, redox, Zn2+) based on 15N and 89Y. These low gamma nuclei present a much wider chemical shift range than 13C, thereby offering higher chemical shift response to changes in the chemical environment. These probes include pH-sensitive hyperpolarized yttrium complexes, hyperpolarized 15N labeled tris(2-pyridylmethyl)amine as 15N-NMR sensor of freely available zinc in biological samples, and H2O2-sensing hyperpolarized 15N-boronobenzyl-4-cyanopyridinium (BBCP).
Contact PI: Zoltán Kovács, Jae Mo Park, A. Dean Sherry
Novel Data Acquisition and Reconstruction Methods
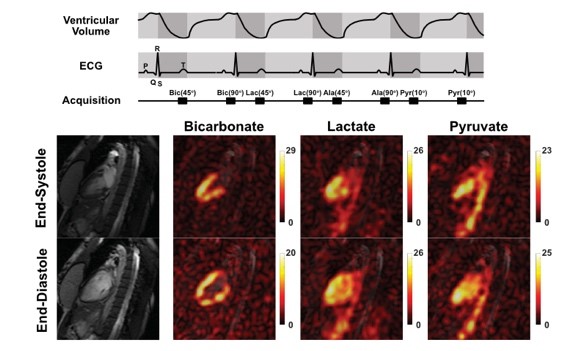
Imaging methods for hyperpolarized compounds need to be rapid due to the real-time nature of in vivo metabolism and the characteristics of hyperpolarized signals. Pulse sequence development and optimization for hyperpolarized signals focus on reliable detection of hyperpolarized products, development of acquisition schemes that capture targeted physiology, and improvement of spatial resolution. AIRC makes available imaging sequences and RF coils for rodents and human brain, cardiac, body, and skeletal muscle. Reconstruction methods that exploit co-registered high-resolution 1H MRI are also developed.
Contact PI: Jae Mo Park
Applications and Clinical Translations
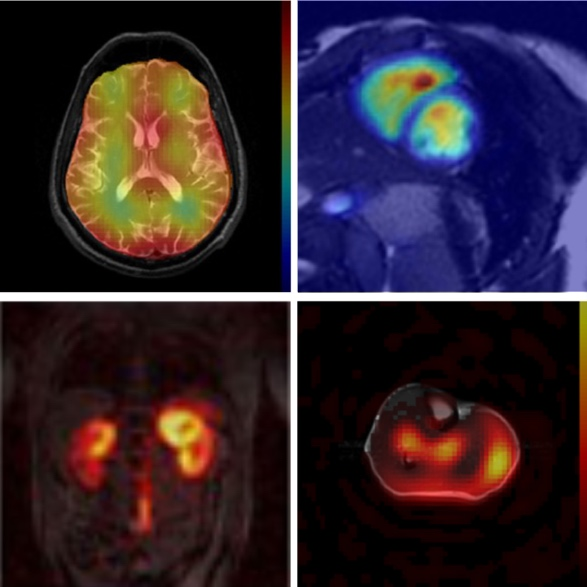
Researchers investigated in-vivo metabolism using hyperpolarized substrates under normal and pathophysiological conditions. Ongoing projects associated with cerebral metabolism include carbohydrate metabolism and branched-chain amino acids metabolism in glioblastoma, and mitochondrial metabolism associated with traumatic brain injury. Investigations of cardiac metabolism showed particular interest in mitochondrial metabolic dysfunction in the myocardium in patients with cardiomyopathies or in cancer patients exposed to anti-cancer treatments. AIRC researchers also study in vivo imaging biomarkers that detect non-alcoholic fatty liver diseases and myopathies.
Contact PI: Jae Mo Park, Craig Malloy
