How gut bacteria become ‘persisters’ to avoid antibiotics
UTSW study shows antibiotic-exposed bacteria developed the ability to die slower, hide inside intestinal cells
DALLAS – Feb. 27, 2024 – A subpopulation of gut bacteria given a commonly used antibiotic became “persisters” that were able to survive without developing true resistance, UT Southwestern Medical Center scientists discovered. Their findings, published in Cell Host & Microbe, could lead to better ways to fight bacterial infections.

“Researchers had hypothesized the existence of these persistent bacteria based on petri dish experiments, but to observe them developing in a living host has never been done,” said Andrew Koh, M.D., Associate Professor of Pediatrics and Microbiology. Dr. Koh co-led the study with Erdal Toprak, Ph.D., Associate Professor of Pharmacology and in the Lyda Hill Department of Bioinformatics. Both are members of the Harold C. Simmons Comprehensive Cancer Center.
Antibiotic resistance is one of modern medicine’s most pressing crises, explained Dr. Koh, with an estimated 2.8 million antibiotic-resistant infections resulting in more than 35,000 deaths a year in the U.S. But how antibiotic resistance develops – particularly within live animal hosts – is unclear. One reason for this is that mice, the quintessential lab model, efficiently clear several antibiotics delivered over set time periods, the way most human patients receive these drugs, causing a mismatch between antibiotic concentrations in mice and humans.
Hoping to observe antibiotic resistance forming in live mice, the researchers developed a mouse model in which antibiotics were delivered continuously through a programmable subcutaneous pump, allowing their serum antibiotic concentrations to remain stable as long as the pump stayed on.

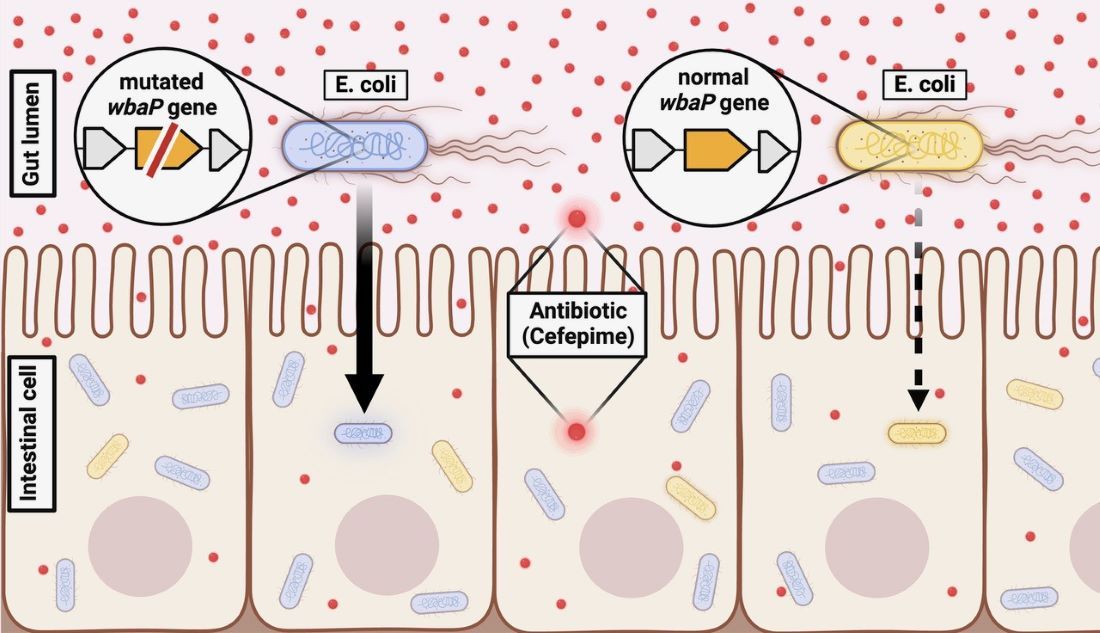
Working with Escherichia coli – a common species of gut bacteria – introduced to “germ-free” mice that didn’t have a natural microbiome, the researchers found that most bacteria were killed by the antibiotic, cefepime. But some of these microbes survived inside intestinal cells and took longer to die when treated with additional antibiotic in petri dishes compared with E. coli isolated from untreated mice.
Genetic analysis showed these persistent bacteria had lost function of a gene called wbaP, which plays an important role in creating a protective coating on the surface of E. coli and some other bacteria species. This loss appeared to be key in allowing the persistent bacteria to invade intestinal cells, where the antibiotic concentration was nearly 20 times lower inside than outside, and in slowing the bacteria’s deaths upon exposure to high antibiotic concentrations.
Such persisters could be the reason why antibiotics sometimes don’t completely clear infections, allowing dangerous antibiotic resistance to develop – a scenario Drs. Toprak and Koh and their colleagues plan to investigate in future studies. The innovative model they developed could be used to examine a variety of questions related to antibiotic resistance in living animals, they added, including whether certain foods, commensal bacteria, other drugs, or autoimmune conditions can affect bacterial survival and resistance upon antibiotic exposure.
Dr. Koh, a physician-scientist, and Dr. Toprak, an experimental biophysicist, noted that their collaboration is a testament to the collegial environment at UTSW, which encourages interdisciplinary partnerships.
“This initiative exemplifies how complex global challenges can be addressed more effectively by uniting diverse scientific fields,” said Dr. Toprak, a Southwestern Medical Foundation Scholar in Biomedical Research.
Other UTSW researchers who contributed to this study include Lora V. Hooper, Ph.D., Chair and Professor of Immunology and Professor of Microbiology and in the Center for the Genetics of Host Defense; Noelle S. Williams, Ph.D., Professor of Biochemistry; Xiaowei Zhan, Ph.D., Associate Professor in the Peter O’Donnell Jr. School of Public Health and the Center for the Genetics of Host Defense; Laura Filkins, Ph.D., Assistant Professor of Pathology; Parastoo Sabaeifard, Ph.D., Senior Research Scientist; Muhammed Sadik Yildiz, Ph.D., postdoctoral researcher; Adam Lyon, graduate student researcher; Laura Coughlin, M.A., Senior Research Associate/Lab Manager; Nicole Poulides, M.S., Research Assistant; Ahmet C. Toprak, medical student; Cassie Behrendt, Research Technician; Xiaoyu Wang, Ph.D., Research Scientist; Marguerite Monogue, Pharm.D., Clinical Pharmacist Specialist; Jiwoong Kim, M.S., Computational Biologist; and Shuheng Gan, M.S., Computational Biologist.
Dr. Hooper holds the Jonathan W. Uhr, M.D., Distinguished Chair in Immunology and is a Nancy Cain and Jeffrey A. Marcus Scholar in Medical Research, in Honor of Dr. Bill S. Vowell. She also is a member of the National Academy of Sciences and a Howard Hughes Medical Institute Investigator.
This study is supported by grants from the National Institutes of Health (K24-AI123163, R01-GM125748, DOD-PR172118, and R01-DK070855), Human Frontiers Science Program Research (RGP0042/2013), The Welch Foundation (I-208-220210327 and I-1874), the Howard Hughes Medical Institute, the UT Southwestern and Children’s Health Cellular and ImmunoTherapeutics Program, and the UTSW Endowed Scholars Program.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 25 members of the National Academy of Sciences, 21 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,100 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in more than 80 specialties to more than 120,000 hospitalized patients, more than 360,000 emergency room cases, and oversee nearly 5 million outpatient visits a year.
