FDA approval of belzutifan culminates 25-year journey at UTSW from gene discovery to a first-in-class drug
DALLAS – Aug. 21, 2021 – A first-in-kind kidney cancer drug developed from laboratory and translational studies conducted at UT Southwestern Medical Center received approval from the Food and Drug Administration, providing a new treatment for patients with familial kidney cancer.
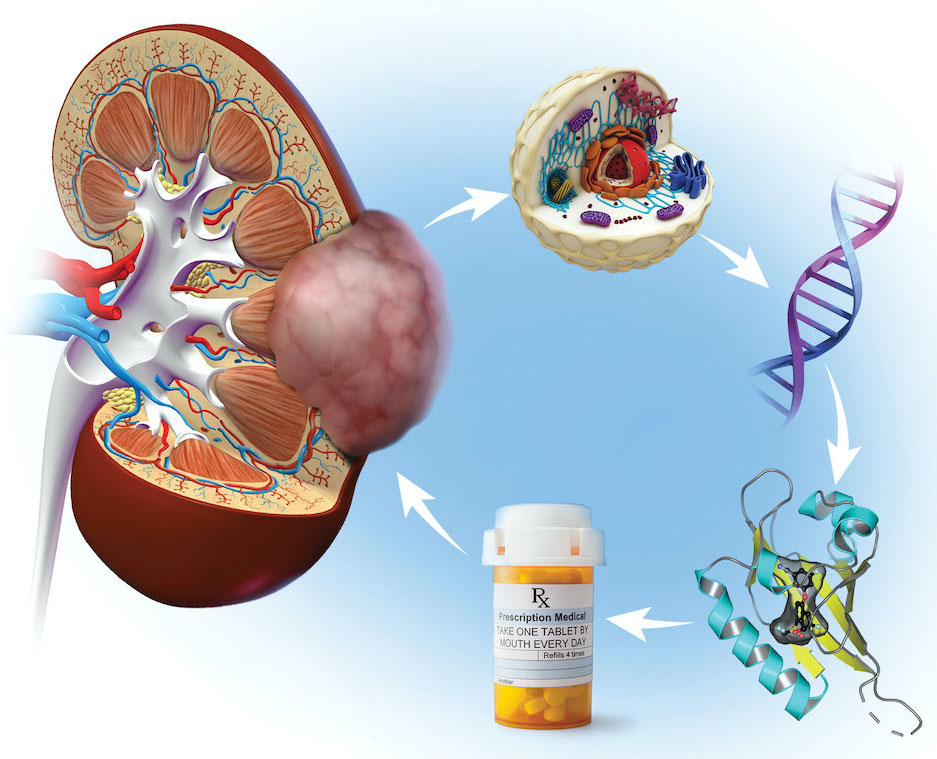
Merck’s belzutifan grew out of the discovery at UT Southwestern of a protein, hypoxia-inducible factor 2-alpha (HIF-2α), that is key to fuel the growth of kidney and other cancers. HIF-2α was discovered by Steven McKnight, Ph.D., Professor of Biochemistry.
“This is an exciting milestone for patients with inherited forms of kidney cancer who are in need of more effective therapies,” said David Russell, Ph.D., Vice Provost and Dean of Research, and Professor of Molecular Genetics, who collaborated in the early stages of the research.
The drug, once called PT2977, was developed based on a backbone discovered by UTSW researchers, with further drug development efforts conducted by a spinoff company named Peloton Therapeutics, which was launched on the UTSW campus and eventually acquired by Merck.
Drs. McKnight and Russell first identified HIF-2α in the 1990s.
HIF-2α was considered undruggable for many years until two more UTSW scientists at the time – Richard Bruick, Ph.D., Professor of Biochemistry, and Kevin Gardner, Ph.D., Professor of Biophysics, who also directs a structural biology center at the City University of New York – did the structural and biochemical work showing that the HIF-2α molecule contains a pocket that is potentially druggable. The two scientists then identified multiple compounds that fit into this pocket and inhibited the activity of HIF-2α.
“The history of belzutifan’s development demonstrates the value of cross-disciplinary collaborations at academic medical centers and how that can translate to new treatments for diseases,” said Dr. Russell. “It also underscores the value of investing in basic science discoveries at the core of advancements in medicine.”
In 2011, several researchers spun off Peloton Therapeutics, and by 2019, when Merck acquired the company, at least three HIF-2α agents were under investigation.

James Brugarolas, M.D., Ph.D., Director of the Kidney Cancer Program at UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center, showed that the drug was effective against kidney cancer.
With funding from a prestigious National Cancer Institute SPORE award, they showed in a publication in Nature in 2016 that the drug was able to inhibit HIF-2α in human kidney tumors transplanted into mice and stop their growth.
This and other studies led to the first clinical trial of PT2385, a precursor to PT2977, which became belzutifan. The trial, which was led by the UTSW Kidney Cancer Program, showed that the drug was well-tolerated and active.
“The approval of belzutifan represents a new paradigm in the treatment of kidney cancer,” said Dr. Brugarolas, Professor of Internal Medicine. “By exclusively targeting HIF-2α, which is essential for kidney cancers but dispensable for normal processes, belzutifan specifically disables cancer cells while sparing normal cells. Belzutifan is the best-tolerated kidney cancer drug today and one suitable for patients with familial kidney cancer. It is a testament to the prowess of designer drugs and carefully chosen targets of which it is a prime example.”
Dr. Brugarolas holds The Sherry Wigley Crow Cancer Research Endowed Chair in Honor of Robert Lewis Kirby, M.D. Dr. McKnight holds the Distinguished Chair in Basic Biomedical Research. Dr. Pedrosa holds the Jack Reynolds, M.D., Chair in Radiology. Dr. Russell holds the Eugene McDermott Distinguished Chair in Molecular Genetics. Disclosures: UT Southwestern and some of its researchers will receive financial compensation, through prior agreements with Peloton, based on belzutifan’s FDA approval.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 25 members of the National Academy of Sciences, 16 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 2,800 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 117,000 hospitalized patients, more than 360,000 emergency room cases, and oversee nearly 3 million outpatient visits a year.
